05.18.2026
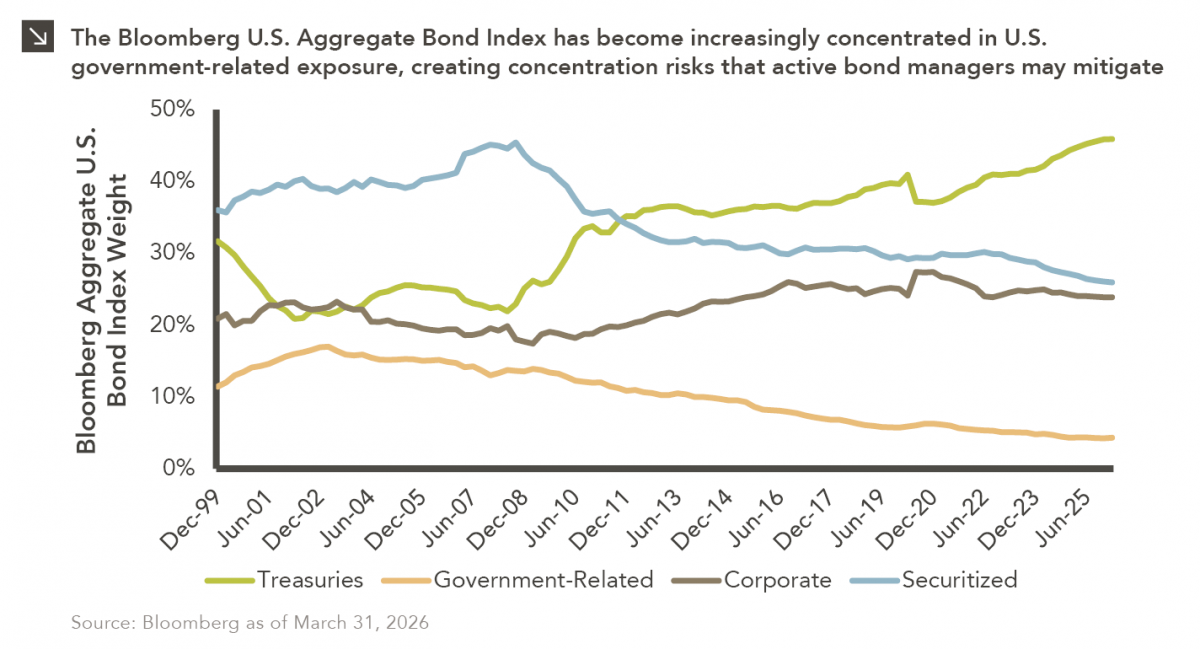
The “Magnificent One”
Over the last few years, equity markets have been defined by a group of stocks often referred to as the…


There are currently 70 coronavirus vaccines being developed by biotech companies around the world. These charts highlight two components of this process.
The left chart shows the clinical trial phases¹ in which these 70 coronavirus vaccine candidates currently reside, with the vaccine candidate from Hong Kong Stock Exchange-listed biopharmaceutical company CanSino Biologics in Phase II, vaccine candidates from NASDAQ-listed Moderna² and NASDAQ-listed Inovio in Phase I, and the rest of the 67 candidates yet to begin Phase I. Of these other 67, notable ones include vaccine candidates from NYSE-listed Pfizer, London Stock-Exchange-listed AstraZeneca, NYSE-listed Johnson & Johnson, Euronext Paris-listed Sanofi jointly with London Stock Exchange-listed GlaxoSmithKline, and Oxford University.
The right chart shows the amount that the U.S. government has invested in coronavirus research so far, with $917 million towards vaccines, $313 million towards therapies, and $24 million towards diagnostics. The $313 million towards therapies is broken out into $152 million for Johnson & Johnson to develop an antiviral drug and $99 million to Regeneron, $25 million to Roche and $37 million to several other biotech companies to develop antibodies. The antivirals and antibodies³ will be used for treating COVID-19 patients and tide us over until a vaccine that can prevent COVID-19 can be developed and distributed.
If a coronavirus vaccine candidate passes Phase III (see endnote), the FDA will review the detailed findings of each of the three phases and approve the treatment if the FDA’s standards are met. However, the process does not stop there. The final phase involves manufacturing, production, and distribution, which may take several months to several quarters. With estimates of late 2020 to mid-2021 for distribution of a coronavirus vaccine cited by investors and the media, the current process for developing, testing, and approving these 70 coronavirus vaccine candidates is certainly on an accelerated path.
Predicting the ultimate success of any singular vaccine is utterly difficult, but with 70 diverse candidates the likelihood that at least one successful vaccine will survive the trial by fire is greater than if only a limited number of vaccines were in development. The 70 vaccine candidates all work differently but have a common trait in that they feature a delivery mechanism for the actual coronavirus spike protein, usually a weakened version of another virus. For example, Oxford University’s vaccine is based on a weakened version of the adenovirus (also known as the common cold virus) from chimpanzees. The genes of the spike protein from the actual coronavirus are spliced into this weakened virus that is altered so that it cannot replicate in humans. This modified virus serves as the vaccine that is injected into the person. Once in cells, this modified virus manufactures the coronavirus spike protein that then prompts the immune system to create antibodies that subsequently fight off the modified virus. The person then is expected to be immune to coronavirus, because if the actual coronavirus were to enter this person’s cells, his or her immune system would be triggered to produce the antibodies to eradicate any virus with this spike protein.
With vaccine development now front and center in terms of a solution to the pandemic-induced recession, our hope is that this evaluation of vaccine progress and the mechanics might be useful for clients vis-à-vis portfolio perspectives, expectations, and decisions in the quarters to come.
Print PDF > Coronavirus Vaccines in Progress
¹ For those not familiar with medical treatment clinical trials, the following summarizes each phase’s timeframe and focus:
Phase I typically lasts several months to a year. Its focus is on whether the drug is safe enough to check for efficacy. Serious side effects will be monitored. Because of the small number of human test subjects — usually 20 to 100 volunteers for Phase I — rare side effects may not be uncovered until phases II or III.
Phase II typically lasts two years. Its focus is on whether the drug has any efficacy. Usually 100 to 300 patients are divided into two or three groups for a controlled experiment by testing the new treatment against other treatments.
Phase III typically lasts several years. Its focus is on the degree of the drug’s therapeutic effect. Usually Phase III involves 300 to 3,000 patients in order to achieve rigorous statistical significance in assessing final levels of efficacy and safety.
² On April 27, 2020, Moderna submitted the application for Phase II, which is expected to start this quarter, and they expect Phase III to start in the fall.
³ Recently Gilead’s antiviral remdesivir was hailed by National Institute of Allergy and Infectious Disease Director Dr. Anthony Fauci as a promising drug that can block COVID-19. In the latest trials, remsdesivir helped patients recover in 11 days versus 15 days with a placebo. Only 8% of patients died taking remdesivir versus 11% of patients who took the placebo. Currently there are several trials to test if remdesivir could stop COVID-19 from replicating. Marquette will continue to monitor remdesivir and other antivirals, as well as the antibody and vaccine candidates, in their progress.
The opinions expressed herein are those of Marquette Associates, Inc. (“Marquette”), and are subject to change without notice. This material is not financial advice or an offer to purchase or sell any product. Marquette reserves the right to modify its current investment strategies and techniques based on changing market dynamics or client needs.
05.18.2026
Over the last few years, equity markets have been defined by a group of stocks often referred to as the…
05.11.2026
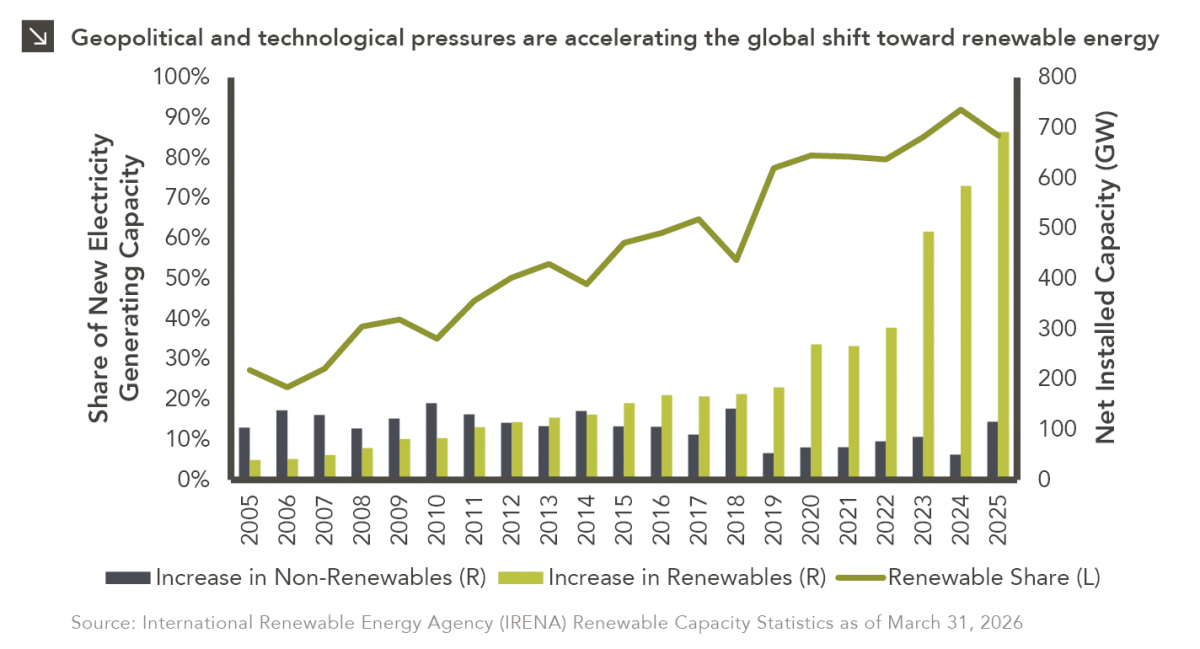
In addition to the humanitarian toll of the conflict in Iran, the world is currently confronting the impact that trade…
05.07.2026
The leadership structure of the Federal Reserve is intentionally designed to promote continuity, independence, and institutional stability across political cycles….
05.04.2026
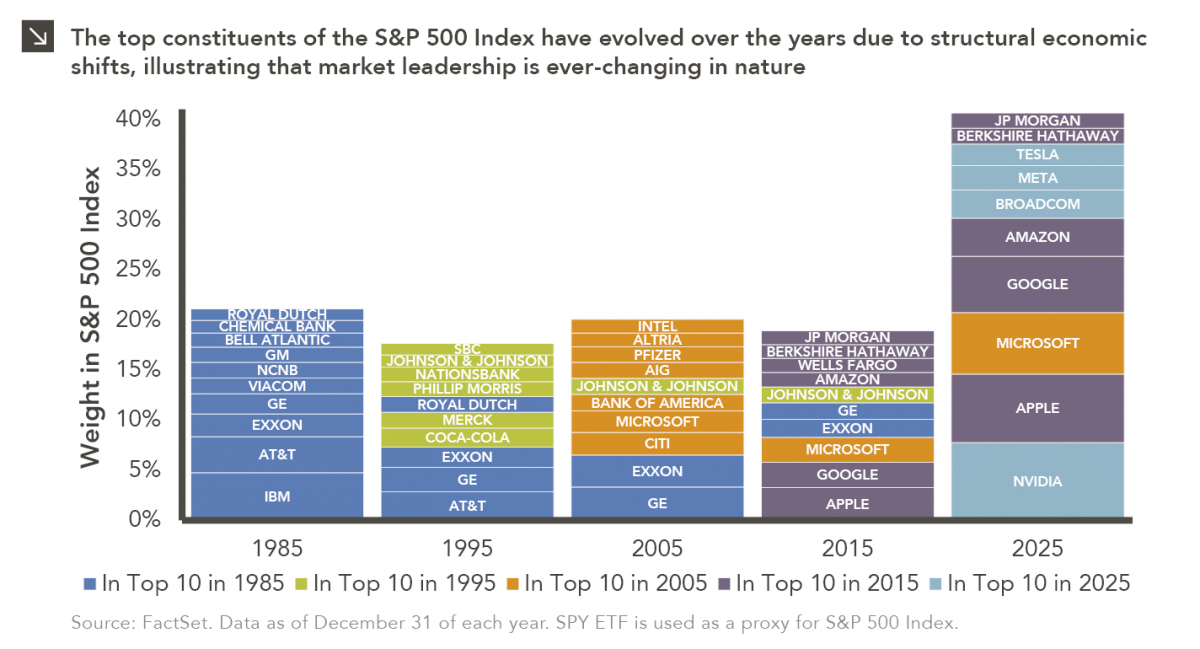
Rooted in medieval Persian Sufi thought, the adage “this too shall pass” speaks to the fleeting and impermanent nature of…
04.27.2026
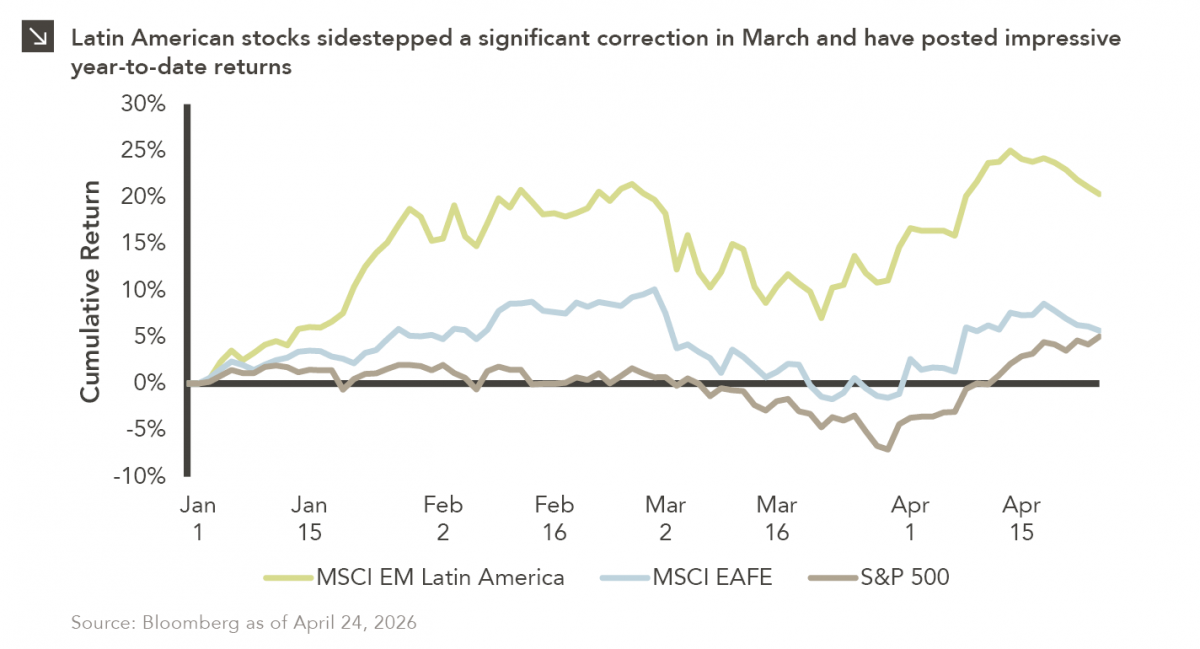
Latin American equity markets have shown remarkable strength in 2026. After a strong start to the year, the MSCI Emerging…
04.23.2026
Diversify. Rebalance. Stay invested. Every one of these letters has concluded with that same advice in some shape or form….
Research alerts keep you updated on our latest research publications. Simply enter your contact information, choose the research alerts you would like to receive and click Subscribe. Alerts will be sent as research is published.
We respect your privacy. We will never share or sell your information.
If you have questions or need further information, please contact us directly and we will respond to your inquiry within 24 hours.
Contact Us >